In 1909, one of Thomsons students, Ernest Rutherford, determined that the positive charge of atoms was located in a central nucleus. The word 'atom' comes from the Greek word for uncuttable. 
By the late 1800’s, John Dalton’s view of atoms as the smallest particles that made up all matter had held sway for about 100 years, but that idea was about to be challenged. Thomson, he received a Nobel Prize for his work in. Atomic theory states that matter is composed of discrete units, called atoms. This module is an updated version of Atomic Theory I. .png)
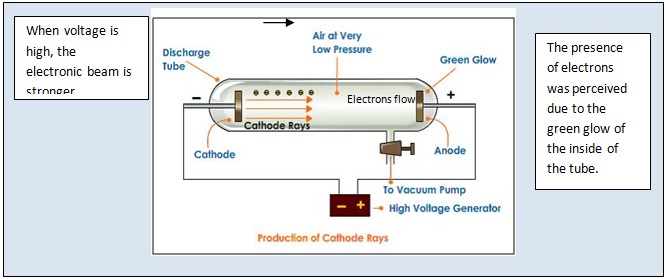
(c) In the cathode ray, the beam (shown in yellow) comes from the cathode and is accelerated past the anode toward a fluorescent scale at the end of the tube. (b) This is an early cathode ray tube, invented in 1897 by Ferdinand Braun. \)) More important, by measuring the extent of the deflection of the cathode rays in magnetic or electric fields of various strengths, Thomson was able to calculate the mass-to-charge ratio of the particles. ThomsonJoseph John Thomson was an English physicist who made major contributions to atomic theory.Known throughout his life as J.J. Thomson produced a visible beam in a cathode ray tube.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
